|
The nested qualitative study will seek to explore the patient’s view of the screening trials, implantation and overall use of SCS. The TRIAL-STIM Study is a randomised controlled trial with a nested qualitative study and economic evaluation aiming to determine the clinical utility of screening trials of SCS as well as their cost-effectiveness. The economic evaluation will take the form of a cost–utility analysis. A nested qualitative study will be carried out in parallel for a total of 30 of the patients recruited in each centre (10 at each centre) to explore their views of the screening trial, implantation and overall use of the SCS device. Secondary outcome measures will include diagnostic accuracy, the proportion of patients achieving at least 50% and 30% pain relief at 6 months as measured on the NRS, health-related quality-of-life (EQ-5D), function (Oswestry Disability Index), patient satisfaction (Patients’ Global Impression of Change) and complication rates. The primary outcome measure is the numerical rating scale (NRS) at 6 months compared between the screening trial and implantation strategy and the implantation-only strategy. Allocation will be stratified by centre and minimised on patient age (≥ 65 or < 65 years), gender, presence of failed back surgery syndrome (or not) and use of high frequency (HF10™) (or not). Subjects will be recruited from the outpatient clinics of the three participating sites and randomised to undergo a screening trial prior to SCS implant or an implantation-only strategy in a 1:1 ratio. A total of 100 adults undergoing SCS implantation for the treatment of neuropathy will be included. The study will take place in three UK centres: South Tees Hospitals NHS Foundation Trust (The James Cook University Hospital) Basildon and Thurrock University Hospitals NHS Foundation Trust and Leeds Teaching Hospitals NHS Trust. The TRIAL-STIM Study is a superiority, parallel-group, three-centre, randomised controlled trial in patients with chronic neuropathic pain with a nested qualitative study and economic evaluation. Any significant local pain should be reported to the doctor.The TRIAL-STIM Study aims to assess the diagnostic performance, clinical outcomes and cost-effectiveness of a screening trial prior to full implantation of a spinal cord stimulation (SCS) device. The site on the back where the leads and electrodes were inserted is usually uncomfortable for a few days. 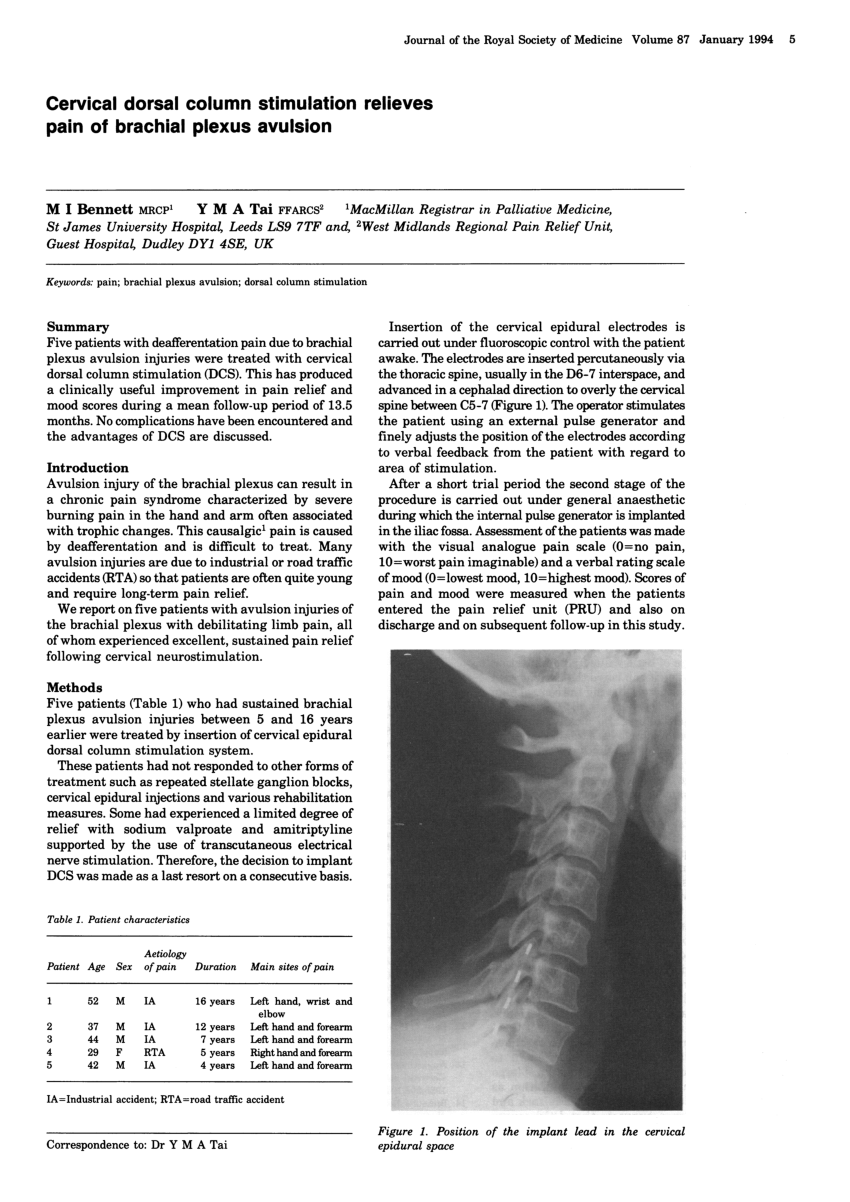
If the pain is not relieved, the doctor should be contacted right away so the device can be reprogrammed. The doctor may ask the patient to track which stimulation settings are used at different times and how well the pain was relieved. 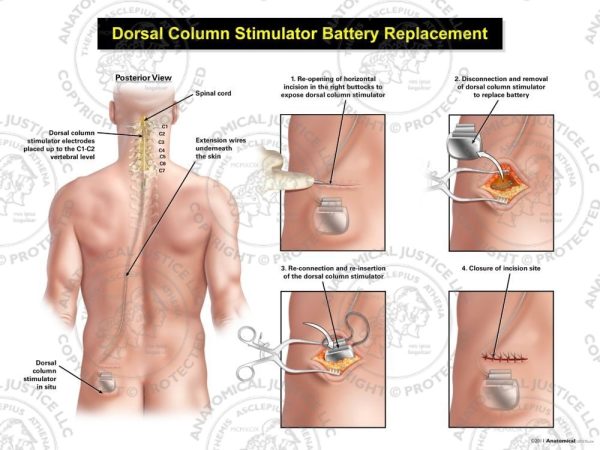
With some models, adjustments are needed for significant body movements, such as getting up or lying down. The controller offers a range of adjustments in the intensity and duration of stimulation. The patient is given the controller and can turn it on to send pulses of current to the electrodes as needed.A hand-held controller to adjust the amount of stimulation is programmed by the doctor based on the earlier feedback from the patient during adjustment of the electrical contacts.The patient is given time to recover from the procedure before going home.See Rechargeable Spinal Cord Stimulators for Chronic Pain The wire connecting to the external neurostimulator is taped to the person’s back during the trial to hold it in place. The leads are connected to an exterior pulse transmitter that the patient wears on a belt.Once the patient has reported on the pain-relief coverage, the patient is again sedated. Devices using newer technology generally avoid the tingling sensation and a wake-up test may not be needed.) Each electrode affects pain in a different area, so doctor-patient communication is crucial in making sure the doctor has adjusted the location of the electrical contacts to cover all the areas in pain. 
(If a low-frequency system is used, the goal will be to cover all painful areas with a slight tingling sensation known as paresthesia. The patient is awakened to provide feedback on the specific areas where pain is relieved by the stimulation, and where pain relief is still required.See Introduction to Diagnostic Studies for Back and Neck Pain In some cases, a small incision may be needed to insert the needle. The needle contains thin, insulated wires, called leads, with electrical contacts attached. Guided by fluoroscopy (a type of X-ray), the doctor inserts a hollow needle into the area around the spinal canal called the epidural space.Local anesthesia is applied to the injection site and sedation may be provided.Procedures vary somewhat with the stimulation device used, but these are the typical steps. The trial period procedure is usually performed in a doctor’s office or a surgical center.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
